TB-500 is a synthetic peptide composed of 43 amino acids and is an analogue of thymosin beta-4 (TB-4), a naturally occurring peptide present in almost all mammalian cells. TB-500 is widely researched for its influence on actin regulation, cellular migration, and tissue repair. Studies conducted in animal models and in vitro environments have demonstrated that TB-500 supports new blood vessel formation, accelerates wound healing, reduces inflammatory responses, and stimulates extracellular matrix development. Ongoing research is also exploring TB-500’s potential role in reducing oxidative stress after spinal cord injuries, enhancing recovery following myocardial infarction, and contributing to various anti-aging mechanisms.
TB-500 Mechanism of Action
TB-500 represents the active functional region of TB-4 and primarily acts as an actin-binding peptide. Actin is a vital structural protein responsible for forming microfilaments within cells. These microfilaments maintain cellular shape, preserve membrane integrity, enable cell movement and migration, and participate in essential stages of cell division. Actin is also a key component of muscle tissue, making it essential for muscle contraction. Without actin, muscular function would not be possible. Actin-binding peptides such as TB-4 bind and protect individual actin monomers, preventing their degradation and ensuring their availability for polymerization into microfilaments when required.
TB-500 (Thymosin Beta-4) Peptide Sequence
TB-500 Peptide Structure
Sequence: Ac-Ser-Asp-Lys-Pro-Asp-Met-Ala-Glu-Ile-Glu-Lys-Phe-Asp-Lys-Ser-Lys-Leu-Lys-Lys-Thr-Glu-Thr-Gln-Glu-Lys-Asn-Pro-Leu-Pro-Ser-Lys-Glu-Thr-Ile-Glu-Gln-Glu-Lys-Gln-Ala-Gly-Glu-Ser
Molecular Formula: C212H350N56O78S
Molar Mass: 4963.4408
CAS Number: 77591-33-4
PubChem: CID 16132341
TB-500 Research
1. TB-500 and Neurologic Function
Research conducted in rat models indicates that TB-500 supports repair and remodeling of both central and peripheral nervous system tissues following injury. While the precise biological pathway remains under investigation, findings suggest that TB-500 stimulates supportive neural cells known as oligodendrocytes, which are responsible for maintaining neuron health[1]. Enhanced activity of these cells has been associated with increased blood vessel formation and neuronal growth in damaged brain regions, resulting in measurable improvements in behavior, motor coordination, and cognitive performance[2].
More recent studies demonstrate that TB-500 may reduce oxidative stress following spinal cord trauma and assist transplanted neural stem/progenitor cells (NSPCs) in surviving long enough to promote spinal tissue regeneration[3]. These results indicate that TB-500 and other TB-4 derivatives may hold promise for future spinal cord injury research. TB-500 could provide valuable insights into recovery pathways that may help restore function in paralyzed regions.
2. TB-500 and Blood Vessel Growth
TB-500 and TB-4 have been shown to strongly stimulate vascular endothelial growth factor (VEGF) expression. VEGF is a key signaling molecule involved in the formation of capillaries, which play a crucial role in wound healing, tissue repair, and even hair growth[4]. Researchers believe TB-500’s involvement in vascular growth extends beyond VEGF stimulation alone. It is thought to contribute to extracellular matrix remodeling, vasculogenesis, angiogenesis, and the differentiation of primitive mesenchymal cells into endothelial cells that line blood vessels. This theory is supported by findings showing that TB-4 deficiency disrupts vascular stability, while external administration enhances capillary development and pericyte recruitment following injury[4].
3. TB-500 and Hair Growth
The association between TB-500 and hair growth was discovered incidentally during laboratory research. Mice genetically lacking TB-4 displayed significantly slower hair regrowth after shaving compared to normal mice. Conversely, mice engineered to overproduce TB-4 demonstrated accelerated hair regrowth. Microscopic examination revealed increased hair shaft density and clustered hair follicles in these mice[5].
4. TB-500 and Antibiotic Synergy
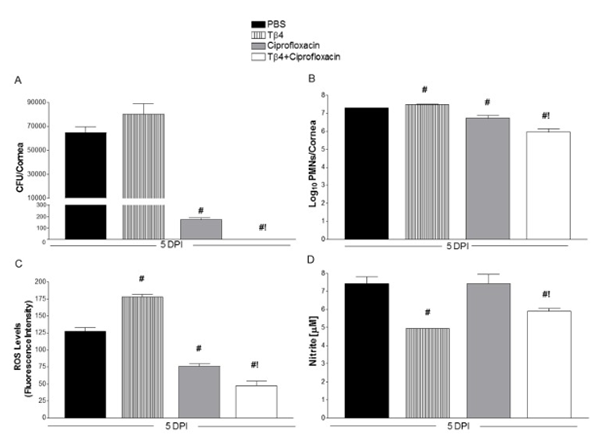
The rise of multidrug-resistant infections has significantly reduced the effectiveness of many existing antibiotics, while the development of new antibiotics remains slow and resource-intensive. Research examining TB-4 and its adjuvant effects offers encouraging insights. Studies involving mice with Pseudomonas aeruginosa eye infections found that combining TB-4 with ciprofloxacin enhanced antibiotic effectiveness, improved healing outcomes, reduced inflammation, and accelerated recovery. After just five days of combination therapy, researchers observed lower bacterial colony counts (CFUs), reduced neutrophil presence, and decreased levels of inflammatory reactive oxygen species[6]. This study was the first to suggest that TB-500-related peptides may help amplify antibiotic efficacy.
TB-500 and Antibiotic Synergy
A. Reduction in bacterial colony forming units (CFUs) after five days of treatment, with undetectable levels observed when ciprofloxacin is combined with TB-4.
B. Neutrophil counts in treated mouse corneas, serving as an indicator of inflammation.
C. Measurement of reactive oxygen species in corneal tissue following treatment.
D. Nitrate concentration analysis from corneal lysates.
Source: PubMed
5. TB-500 and Cardiovascular Health
Over twenty years of research suggest that TB-4 and its derivatives offer multiple benefits within the cardiovascular and renal systems. Although the exact biological mechanisms are not fully understood, evidence indicates several contributing factors. TB-500 supports the development of collateral blood vessels, aiding both disease prevention and functional recovery. It also promotes endothelial cell migration and enhances cardiomyocyte survival following myocardial infarction. Additionally, TB-500 appears to interact with other natural signaling molecules to reduce inflammation and limit fibrosis (scar tissue formation)[7].
Recent studies involving hydrogels composed of collagen and TB-4 have shown that the peptide enhances angiogenesis and epicardial heart cell migration. These effects improve recovery following ischemic events and help reduce long-term complications by minimizing scar formation[8].
6. TB-500 and Neurodegenerative Diseases
Progress toward effective treatments for neurodegenerative conditions such as Alzheimer’s disease and prion disorders has been limited. However, recent research examining TB-4’s interaction with immune responses to prion proteins suggests that the peptide enhances autophagy[9]. Autophagy is a key protective mechanism within the central nervous system, and TB-4’s ability to strengthen this process represents a promising step toward potential therapeutic strategies.
7. TB-500 Has Wide Application
Due to its fundamental involvement in cellular structure and function, TB-500 influences a wide range of tissues throughout the body. This broad biological role has led to extensive research across numerous medical fields. From cardiovascular and neurological studies to investigations into antibiotic enhancement, TB-500 remains one of the most actively researched peptides today and is expected to continue attracting scientific interest for years to come.
TB-500 has demonstrated minimal side effects, low oral bioavailability, and strong subcutaneous bioavailability in mouse studies. Dosages used in animal research do not translate directly to human use. TB-500 offered by Sana is intended strictly for educational and scientific research purposes and is not approved for human or animal consumption. Purchase is limited to qualified and licensed researchers.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Allan L. Goldstein, MD, Allan L. Goldstein is professor and Catharine B. & William McCormick Chair of the department of Biochemistry and Molecular Biology at The George Washington University School of Medicine and Health Sciences, where he has served since 1978. Thymosins were discovered in the mid 1960’s, when Allan Goldstein from the Laboratory of Abraham White at the Albert Einstein College of Medicine in New York studied the role of the thymus in development of the vertebrate immune system. He is a world-renowned authority on the thymus gland and the workings of the immune system, and co-discoverer of the thymosins. Dr. Goldstein is the author of over 400 scientific articles in professional journals, the inventor on more than 15 U.S. Patents, and the editor of several books in the fields of biochemistry, biomedicine, immunology and neuro-science. He is on the editorial boards of numerous scientific and medical journals and has been a consultant to many re-search organizations in industry and government; co-founder of The Institute for Advanced Studies in Aging and Geriatric Medicine, a non-profit research and educational institute; a member of the Board of Trustees of the Albert Sabin Vaccine Institute; and serves as the Chairman of the Board of RegeneRx Biopharmaceuticals. Dr. Goldstein received his B.S. from Wagner College in 1959 and his M.S. and Ph.D. from Rutgers University in 1964. He served as a faculty member of the Albert Einstein College of Medicine from 1964 to 1972, and moved to the University of Texas Medical Branch in Galveston in 1972 as professor and director of the division of Biochemistry.
Allan L. Goldstein, MD is being referenced as one of the leading scientists involved in the research and development of TB-500 and other Thymosins. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Goldstein is listed in [11] under the referenced citations.
Referenced Citations
Cheng, F. Kuang, H. Zhang, G. Ju, and J. Wang, “Beneficial effects of thymosin β4 on spinal cord injury in the rat,” Neuropharmacology, vol. 85, pp. 408–416, Oct. 2014. [PubMed]
Chopp and Z. G. Zhang, “Thymosin β4 as a restorative/regenerative therapy for neurological injury and neurodegenerative diseases,” Expert Opin. Biol. Ther., vol. 15 Suppl 1, pp. S9-12, 2015. [PubMed]
Li, Y. Wang, X. Hu, B. Ma, and H. Zhang, “Thymosin beta 4 attenuates oxidative stress-induced injury of spinal cord-derived neural stem/progenitor cells through the TLR4/MyD88 pathway,” Gene, vol. 707, pp. 136–142, May 2019. [PubMed]
N. Dubé and N. Smart, “Thymosin β4 and the vasculature: multiple roles in development, repair and protection against disease,” Expert Opin. Biol. Ther., vol. 18, no. sup1, pp. 131–139, 2018. [PubMed]
Philp, S. St-Surin, H.-J. Cha, H.-S. Moon, H. K. Kleinman, and M. Elkin, “Thymosin beta 4 induces hair growth via stem cell migration and differentiation,” Ann. N. Y. Acad. Sci., vol. 1112, pp. 95–103, Sep. 2007. [PubMed]
W. Carion et al., “Thymosin Beta-4 and Ciprofloxacin Adjunctive Therapy Improves Pseudomonas aeruginosa-Induced Keratitis,” Cells, vol. 7, no. 10, Sep. 2018. [PubMed]
M. Kassem, S. Vaid, H. Peng, S. Sarkar, and N.-E. Rhaleb, “Tβ4-Ac-SDKP pathway: Any relevance for the cardiovascular system?,” Can. J. Physiol. Pharmacol., pp. 1–11, Mar. 2019. [PubMed]
D. Shaghiera, P. Widiyanti, and H. Yusuf, “Synthesis and Characterization of Injectable Hydrogels with Varying Collagen–Chitosan–Thymosin β4 Composition for Myocardial Infarction Therapy,” J. Funct. Biomater., vol. 9, no. 2, Mar. 2018. [PubMed]
-J. Han, S. Kim, and J. Kwon, “Thymosin beta 4-Induced Autophagy Increases Cholinergic Signaling in PrP (106-126)-Treated HT22 Cells,” Neurotox. Res., Dec. 2018. [PubMed]
Song, Ran & Choi, Hyun & Yang, Hyung-In & Yoo, Myung & Park, Yong-Beom & Kim, Kyoung. (2012). Association between serum thymosin β4 levels of rheumatoid arthritis patients and disease activity and response to therapy. Clinical rheumatology. 31. 1253-8. 10.1007/s10067-012-2011-7. [Research Gate]
Philp, D., et al. “Thymosin β4 Promotes Angiogenesis, Wound Healing, and Hair Follicle Development.” Mechanisms of Ageing and Development, vol. 125, no. 2, Feb. 2004, pp. 113–115, 10.1016/j.mad.2003.11.005. [PubMed]
ALL ARTICLES AND PRODUCT INFORMATION ON THIS WEBSITE ARE PROVIDED FOR EDUCATIONAL AND INFORMATIONAL PURPOSES ONLY.
The products available on this website are intended solely for in-vitro laboratory research. In-vitro studies are conducted outside of living organisms. These products are not approved medications or drugs and have not been evaluated by the FDA for the prevention, treatment, or cure of any disease or medical condition. Introduction into humans or animals is strictly prohibited by law.
 Storage & Stability
Storage & Stability
-
Store lyophilized TB-500 at -20°C for long-term preservation
-
Reconstituted solutions should be stored refrigerated according to laboratory protocols
-
Protect from heat, light, and moisture
-
Avoid repeated freeze-thaw cycles
-
Handle using sterile laboratory techniques
Proper storage conditions are essential to maintain peptide stability and research integrity.










