Ipamorelin is a synthetic peptide widely researched for its selective interaction with growth hormone–releasing pathways. It is commonly studied in controlled research settings for its potential role in endocrine signaling, metabolic regulation, and biological response mechanisms.
At Sana, Ipamorelin is manufactured and handled under strict quality control procedures to ensure consistency, purity, and reliability. Each batch is tested to meet research-grade standards, making it suitable for laboratory-based studies and educational research applications.
This compound is valued in research environments due to its targeted mechanism and stability profile.
Biochemical Characteristics
Source: PubChem
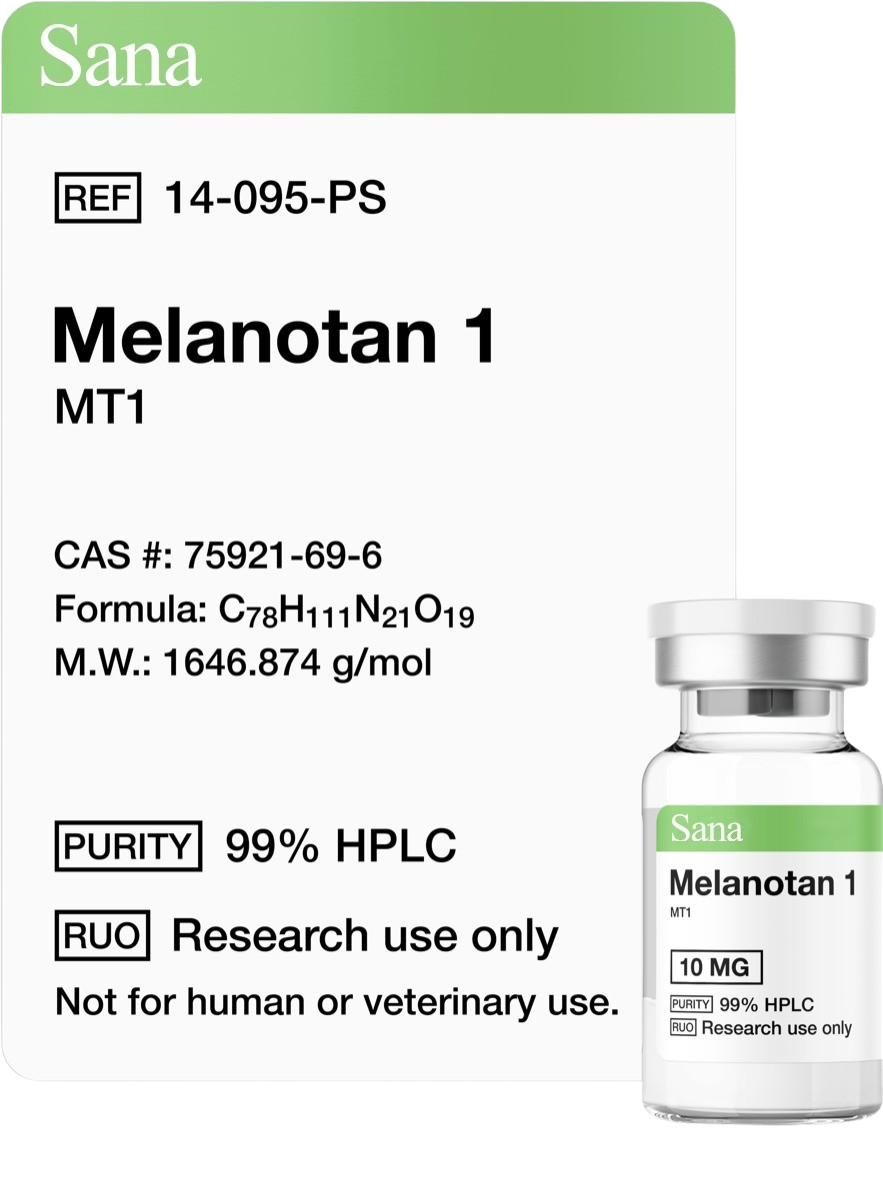
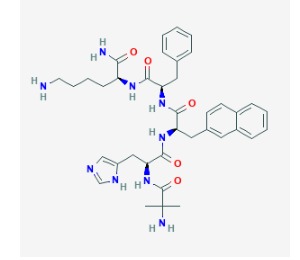
Peptide Sequence: Aib-His-D-2Nal-D-Phe-Lys
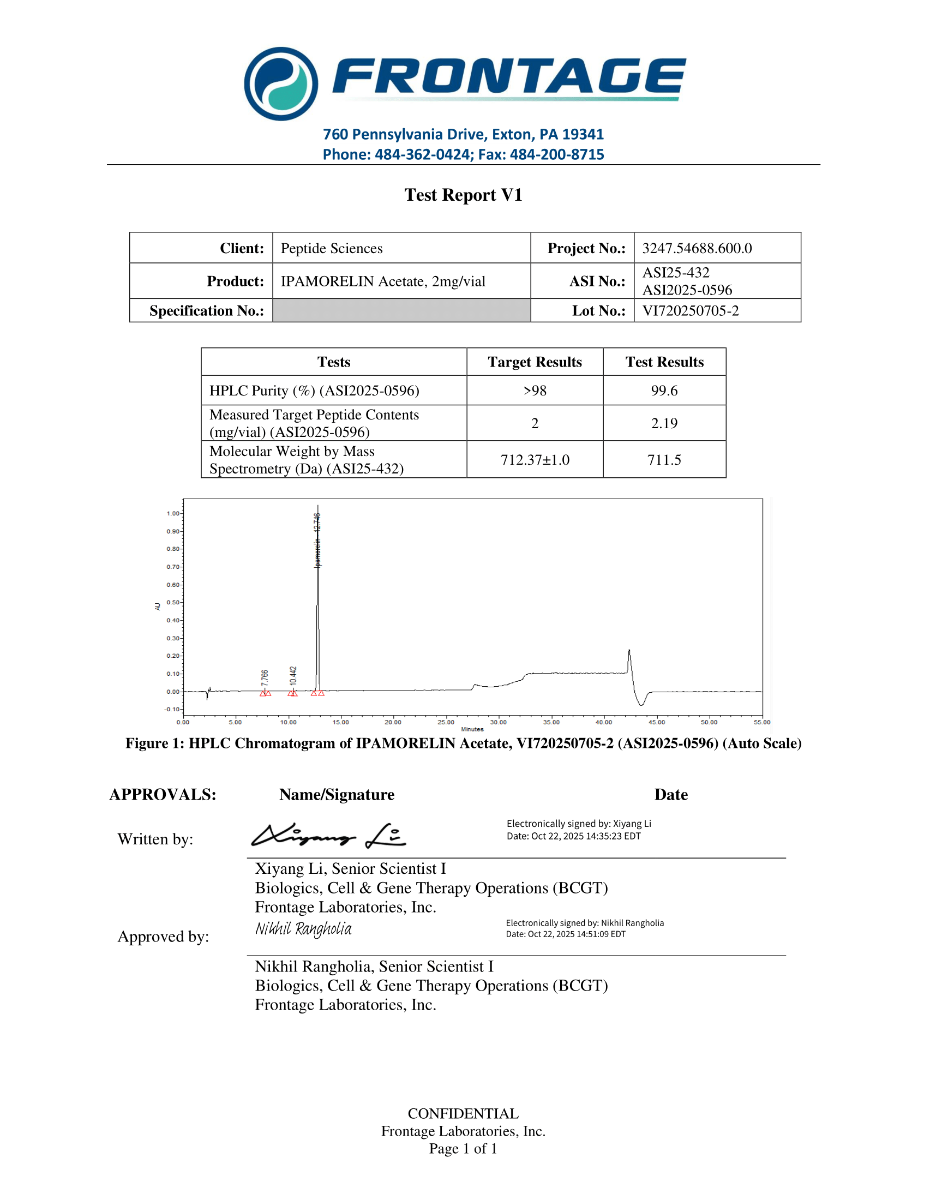
Molecular Formula: C38H49N9O5
Molecular Weight: 711.868 g/mol
PubChem CID: 9831659
CAS Number: 170851-70-4
The presence of non-proteinogenic residues is leveraged in experimental design to evaluate receptor-binding selectivity and metabolic stability in model systems. Ipamorelin is commonly synthesized via solid-phase peptide synthesis and characterized using chromatographic and spectrometric methods.
Research Applications
Ipamorelin is supplied exclusively for laboratory research and is used in preclinical or in-vitro studies involving:
- GHSR-1a binding affinity and selectivity assays
- Second-messenger signaling analysis, including adenylate cyclase/cAMP and calcium-dependent pathway readouts
- Receptor–ligand SAR investigations and comparator studies versus other ghrelin mimetics
- Endocrine-axis modeling in animal systems as a receptor-selective pharmacology tool compound
- Experimental models evaluating GI motility signaling, pancreatic islet signaling, and metabolic pathway endpoints in controlled preclinical contexts
All applications are restricted to non-clinical experimental contexts.
Pathway / Mechanistic Context
Ipamorelin is used to probe GHSR-1a–linked GPCR signaling in which receptor engagement can trigger calcium mobilization and modulation of adenylate cyclase activity, leading to downstream phosphorylation events and transcriptional responses. In vitro and animal models have used ipamorelin to evaluate how selective GHSR activation influences second-messenger dynamics and pathway coupling across Gq– and Gs-associated signaling nodes.
Due to reported selectivity in preclinical systems, ipamorelin is frequently incorporated as a mechanistic comparator in receptor pharmacology research to help differentiate GHSR-mediated signaling from broader secretagogue activity observed with less selective ligands.
Preclinical Research Summary
Glucocorticoid-Associated Signaling Models
Rodent studies have evaluated ipamorelin in experimental systems modeling glucocorticoid-associated alterations in bone, muscle, and nitrogen metabolism. Reported endpoints include osteoblastic activity markers, bone formation indices, and nitrogen-balance–related measurements under controlled laboratory conditions[2], [3], [4].
Pancreatic Islet and Metabolic Signaling
In isolated pancreas preparations and diabetic rodent models, ipamorelin has been investigated for effects on insulin-release–associated mechanisms, including experimental observations related to calcium-dependent signaling in pancreatic islet cells within defined study designs[5].
Gastrointestinal Motility Models
Preclinical rodent models of postoperative ileus have been used to examine ghrelin-mimetic signaling on gastrointestinal motility. These studies assess parameters such as gastric emptying and intestinal transit distribution using radiolabeled markers to support mechanistic exploration of enteric signaling pathways[7].
Source: PubMed
Molecular Imaging Probe Development
Due to receptor selectivity and synthetic accessibility, ipamorelin has been explored as a scaffold for peptidomimetic derivatives in molecular imaging research. In-vitro feasibility studies have evaluated radiolabeled analogues for ghrelin receptor visualization to support experimental imaging system development[8].
Form & Analytical Testing
Ipamorelin is supplied as a synthetic peptide intended for laboratory handling. Identity and purity are verified using high-performance liquid chromatography (HPLC) and mass spectrometry (MS). Batch-specific analytical documentation supports reproducibility and experimental consistency.
Article Author
The above scientific material was researched, compiled, and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a Bachelor of Science degree in molecular biology. The content reflects a synthesis of publicly available preclinical literature and biochemical research intended solely for laboratory reference.
Scientific Journal Author
David E. Beck, MD is a contributing author to peer-reviewed research examining ghrelin mimetics, including ipamorelin, within controlled experimental and clinical research environments. His academic work focuses on gastrointestinal physiology and surgical outcomes, providing foundational data relevant to mechanistic and translational research models.
David E. Beck, MD is cited strictly for academic attribution purposes. No endorsement, affiliation, or commercial relationship is implied or expressed between this researcher and any product offering. Reference is provided solely to acknowledge published scientific contributions. David E. Beck, MD is listed in [6] under the referenced citations.
Referenced Citations
Raun et al., “Ipamorelin, the first selective growth hormone secretagogue,” Eur. J. Endocrinol., vol. 139, no. 5, pp. 552–561, Nov. 1998. [PubMed]
B. Andersen, K. Malmlöf, P. B. Johansen, T. T. Andreassen, G. Ørtoft, and H. Oxlund, “The growth hormone secretagogue ipamorelin counteracts glucocorticoid-induced decrease in bone formation of adult rats,” Growth Horm. IGF Res., vol. 11, no. 5, pp. 266–272, Oct. 2001. [PubMed]
Svensson et al., “The GH secretagogues ipamorelin and GH-releasing peptide-6 increase bone mineral content in adult female rats,” J. Endocrinol., vol. 165, no. 3, pp. 569–577, Jun. 2000. [PubMed]
K. Aagaard et al., “Growth hormone and growth hormone secretagogue effects on nitrogen balance and urea synthesis in steroid treated rats,” Growth Horm. IGF Res., vol. 19, no. 5, pp. 426–431, Oct. 2009. [PubMed]
Adeghate and A. S. Ponery, “Mechanism of ipamorelin-evoked insulin release from the pancreas of normal and diabetic rats,” Neuro Endocrinol. Lett., vol. 25, no. 6, pp. 403–406, Dec. 2004. [PubMed]
E. Beck et al., “Prospective, randomized, controlled, proof-of-concept study of the Ghrelin mimetic ipamorelin for the management of postoperative ileus in bowel resection patients,” Int. J. Colorectal Dis., vol. 29, no. 12, pp. 1527–1534, Dec. 2014. [PubMed]
Greenwood-Van Meerveld et al., “Efficacy of ipamorelin, a ghrelin mimetic, on gastric dysmotility in a rodent model of postoperative ileus,” J. Exp. Pharmacol., vol. 4, pp. 149–155, Oct. 2012. [PubMed]
M. Fowkes et al., “Peptidomimetic growth hormone secretagogue derivatives for positron emission tomography imaging of the ghrelin receptor,” Eur. J. Med. Chem., vol. 157, pp. 1500–1511, Sep. 2018. [Science Direct]
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.
Storage & Handling
-
Store in a cool, dry environment
-
Protect from light and moisture
-
Handle using standard laboratory protocols
-
Keep container tightly sealed when not in use